News
Key Protein for Plant-Fungal Symbiosis Discovered
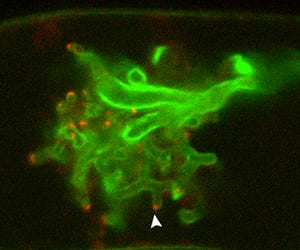
A live arbuscule, forming within a plant cell, lights up green under a confocal microscope. The EXO70I proteins show up as red dots next to the tips of the branches.
Researchers in the laboratory of BTI Professor Maria Harrison have discovered a new protein, called EXO70I, which is required by plant roots to form a successful symbiotic relationship with soil fungi.
In a paper published August 17 in Current Biology, Harrison, with postdoctoral researchers Xinchun Zhang, Nathan Pumplin and Sergey Ivanov, report that plants use EXO70I to form a membrane around the fungus in arbuscular mycorrhizal (AM) symbioses, beneficial associations where plants receive phosphate from fungi in exchange for carbohydrates.
When a tendril of fungus, called a hypha, begins to colonize a root cell, it breaches the cell wall and begins to form a branched structure called an arbuscule. As it grows, the plant cell deposits a membrane around the fungus, called the periarbuscular membrane. The membrane is full of transporters that take up phosphate from the fungus. The researchers think that EXO70I serves as a docking station on the root cell membrane, where bubbles of membrane and transport proteins, synthesized inside the plant cell, can attach and fuse with the cell membrane to then form a layer around the fungus.
“All plants that have the capacity to develop AM symbiosis have a unique EXO70 gene, called EXO70I, that is essential for accommodation of the arbuscule and therefore essential for the symbiosis to function,” said Harrison. “Basically without this protein, the fungus can’t live inside the plant cells and consequently, the symbiotic association does not develop.”
Previous studies have explored the function of the EXO70 protein in yeast, finding that it is part of exocytosis, where membrane-bound, cellular packages fuse to the surface of the cell to expel their contents. Specifically, EXO70 acts as a tethering point for the package on the cell membrane. Plants have the same machinery but have multiple EXO70 proteins—39 in rice and 26 in the experimental plant, Medicago truncatula— which may each be used in specific situations, such as membrane development during symbiosis.
Harrison and her laboratory members first identified EXO70I as an important protein in AM formation by looking for proteins that were turned on at the same time as another protein required for the symbiosis, called Vapyrin.
When they tested its activities, they found that EXO70I is not required for building the periarbuscular membrane around the “trunk” of the arbuscule, where the fungus enters the plant cell, only the branches. They have not yet found the protein responsible for putting a membrane around the trunk, “so that part of the puzzle is still to be determined,” said Harrison.
In future work, Harrison plans to identify other proteins that work in concert with EXO70I to develop the symbiosis, and to look for other EXO70 proteins that may build other portions of the periarbuscular membrane.
This discovery further illuminates the complex interrelationships between plant roots and AM fungi. Ultimately, researchers may use this information to enhance phosphate delivery to plants through the symbiosis and reduce the need for added fertilizers.

